Alan Wade
What do bees use to reorganise themselves when they are dropped off a frame outside the hive or the hive topples over? How do bees maintain cohesion when migrating as a swarm? Why do bees raise their abdomens and fan vigorously at their hive entrance or when bees enter a swarm box? And how do drones communicate with each other and attract virgin queens to congregation areas well away from their natal domicile?
And why do bees flit from one dandelion bloom to another with pinpoint accuracy never returning to flowers just visited? How to foragers recognise and remain faithful to a particular flower type?
We might classify these responses into two groups: pheromones produced by bees that allow them to recognise other bees and chemical scents produced by flowers.
In this follow up story on bee communication we examine the chemical sensing mechanisms that bees employ exterior to the hive. Some of these, such as those produced by worker Nasonov glands are used to help bees congregate when stranded from the hive and that fanning bees employ when entering a new hollow. The same Nasonov gland in the giant honey bee (Apis dorsata) produces alarm pheromones employed in aggressive defence.
Smelling the roses
Spray lavender water into a hive and bees will emerge en masse, fly across wind and head up wind to the lavender patch all the time sensing an increasing gradient of lavender oil comprising primarily linalool, linalyl acetate, lavandulyl acetate, terpinen-4-ol, lavandulol, 1,8-cineole and camphori. Presumably bees can discriminate different species, and potentially different patches, of lavender (Lavendula spp) as each would have a different reward and different mix of chemicals.
In examining the extrinsic sensory world of the bee outside the hive we learn that bees rely on a whole more on what they smell than what they see to gain orientation. This world is entirely different to that of the darkened confines of the nest. While visual signalling would appear to dominate successful navigation of the fifty or so square kilometres a colony of bees patrol, when it comes to targeting sources of nectar, pollen, propolis and water they are likely almost largely reliant on their sense of smell?
Sniffer radar
A good starting point to examine these mysteries is to examine the general morphology of the bee. Insects such as bees have extraordinarily sensitive ‘sniffer organs’, things more accurately termed antennae or colloquially feelers. Each bee antennae has 170 individual chemoreceptor cells: each locus can detect a single molecule of a volatile substance. Large mushroom bodies in the forebrains of invertebrates that process olfactory signals certainly suggests that bees have an acute sense of smell.
There are several categories of chemicals that invertebrates such as insects can recognise with remarkable ease. We know that dung beetles can detect a cow dropping like a homing pigeon and that dipterans such as mosquitoes and flies — the filth insects — know where their next meal is. Mosquitoes can detect thermal heat and carbon dioxide gradients, flies excrement within seconds of deposition. And for bees?
Inside the hive, trophallaxis — exchange of food — is the primary means of transmission of pheromones that control collective behaviour. Volatiles, both those generated by flowers and those manipulated or manufactured by bees themselves are also detected by antennation. Prominent amongst the natural volatiles are the cyclic and acyclic terpenes (ten and fifteen carbon units built on the basic five-carbon building block isoprene) such as isomers of limonene (oranges), camphor (from camphor laurel) and pinene (pine tree oil) and simple aromatics such as eugenol (oil of cloves), cinnamaldehyde (cinnamon) and coumarin (principal constituent of new mown hay) (Figure 4).
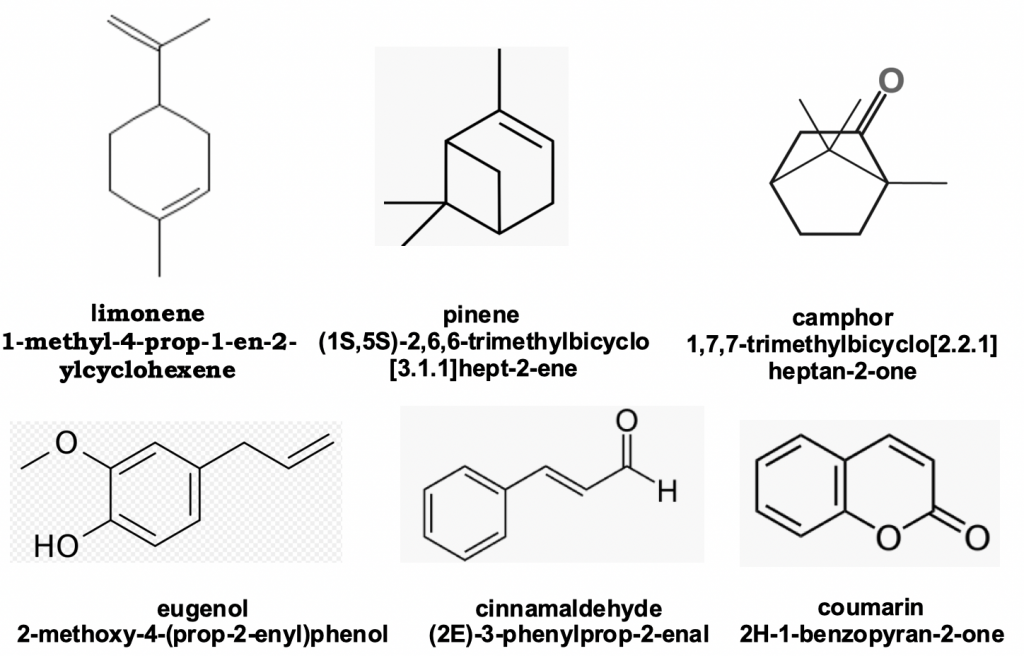
Figure 4 Common plant volatiles: cyclic monoterpenes and aromatics oils
The Nasonov gland
Bees also spray a natural array of terpenes through their Nasonov gland located under the seventh dorsal abdominal tergite (Figure 5). The Nasonov gland was first described in 1882 by the Russian zoologist Nikolai Viktorovich Nasonov (1855 – 1939)ii.
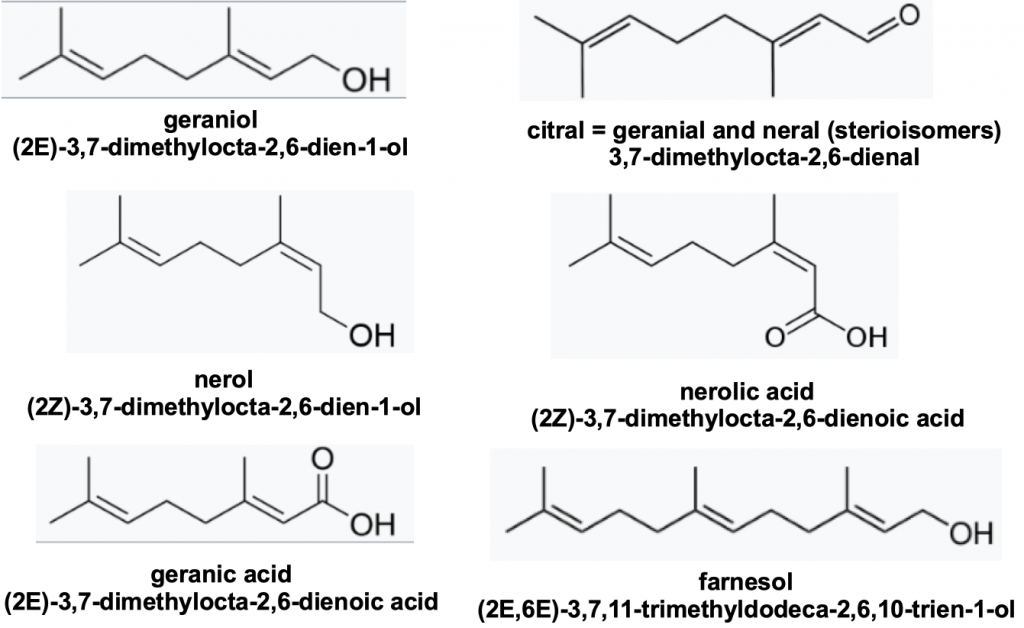
Figure 5 Nasonov gland volatiles [essential oils] in Apis mellifera: geraniol 100 parts, geranial 1 part, neral 1 part, nerol 1 part, nerolic acid 75 parts, geranic acid 12 parts and farnesol 50 partsiii
In a follow up study Williams and coworkers were able to show that honey bees were attracted to each of these terpenes alone.
Interestingly geraniol is the base monoterpene natural product from which other terpenes are routinely synthesised. I’m emphatically also not an invertebrate neurobiologist so will not hazard a guess as to how bees detect and process signalling from these chemical mixtures and really make sense of these chemicals from having handled some them in the laboratory and, of course, sniffing geraniums.
However bees recognise many chemicals innately – they employ individual olfactory receptors on their antennae – including learning how to detect a wide range of naturally occurring volatiles. Chemical signalling is not only of importance to bees themselves: it is being increasingly employed to act as swarm luresiv and to detect and attract exotic species of honey bee such as the Javanese strain of the Asian Honey Beev threatening Australian and global beekeeping enterprises.
Nasonov terpene signatures differ across the honey bee genus. Only neral has been detected in Apis indica (syn Apis cerana indica)vi though a mixture of linalool, linalool oxide and citral (geranial and neral) has been found in the Japanese race of Apis ceranavii (Figure 6).
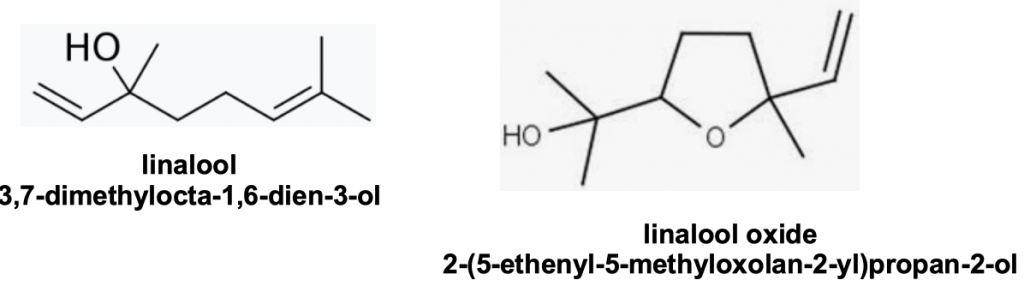
Figure 6 Nasonov pheromone in Apis cerana japonica
There are a number of signalling chemical groups impacting on behavior of bees outside the hive, alarm pheromones used in defence, footprint pheromones used to mark hive entrancesviii and flower marker pheromones. Not surprisingly different species of honey bee employ their pheromones in various ways to achieve different ends. For example the Nasonov pheromones in in Apis dorsata are less associated with aggregation behaviour and more with pheromone release and abdomen waggling behaviour associated with defenceix.
Alarm pheromone
In Apis mellifera alarm pheromone is produced by both mandibular and sting Koschevnikov glandsx,xi(Figure 7): the mandibles release 2-heptanone while stinging bees release a range of alcohols and esters. One of these esters, isoamyl acetate, is a key constituent of ripe bananas – the familiar smell of a satchel in the school locker room. A good rule of thumb is avoid packing bananas for that visit to the apiary. However we should not delude ourselves and assume we have anything like a good grasp on something as ostensibly simple as sting pheromone chemical constituents and what constitutes colony defence response.
Cassier, Tel-Zur and Lenskyxii discuss the role when both sting and associated Koschevnikovxiii glands act in synergy and the vast array of aliphatic and aromatic hydrocarbons, alcohols, aldehydes and ketones, esters and carboxylic acids – some 46 or so – that have been identified and whose individual function if not all are understood.
Camargos and coworkers found twenty three Koschevnikov gland (the gland associated with the worker bee sting apparatus) compounds, identifying twenty one: hexan-1-ol, isoamyl acetate [isopentyl acetate], hexyl acetate, octan-2-one [n-hexyl methyl ketone], nonan-2-one [methyl heptyl ketone], nonan-2-yl acetate [1-methyloctyl acetate], 2-decen-1-ol [(E)-dec-2-en-1-ol], 2-dodecanyl acetate, pentadecyl acetate, 9-octadecanal [(E)-octadec-9-enal], oleic acid [(Z)-octadec-9-enoic acid], nonadecan-1-ol [nonadecyl alcohol], (Z)-9-icosen-1-ol [gadoleyl alcohol], (Z)-9-tricosene, tricosane, 2-methyltetracosane, 7-hexylicosane, heptacosane, pentatriacontane and hexatriacontane.
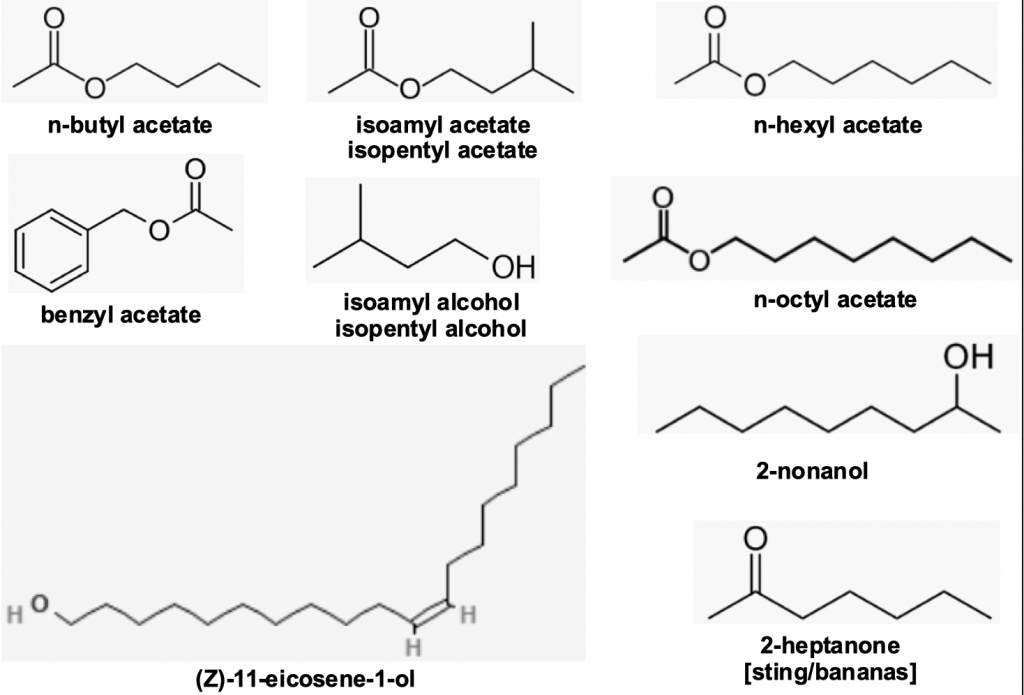
Figure 7 Alarm pheromones: a sample of bee sting gland volatiles
Collisonxiv explains that there are genetic differences between foragers (gatherers) and guard (alarm pheromone emitting) and soldier (harassers of predators) castes of worker bees so clearly defense behaviour is a very complex matter.
Drone congregation pheromones
These comprise a mixture of forty seven lipids, saturated, unsaturated and methyl branched fatty acids and several long chain alcoholsxv (Figure 8) identified from extracts of drone mandibular glands. Again this looks like an unholy chemical concoction though most are common constituents of animal fats. Think of them particularly the saturated fellows – the ones without the double bonds – as a fairly exotic mix of unhealthy long chain fatty acids. It is hard to conceive how these essentially non volatile acids might act on the wing but they appear to affect the behaviour of other drone, but not worker, bees inside the hive.
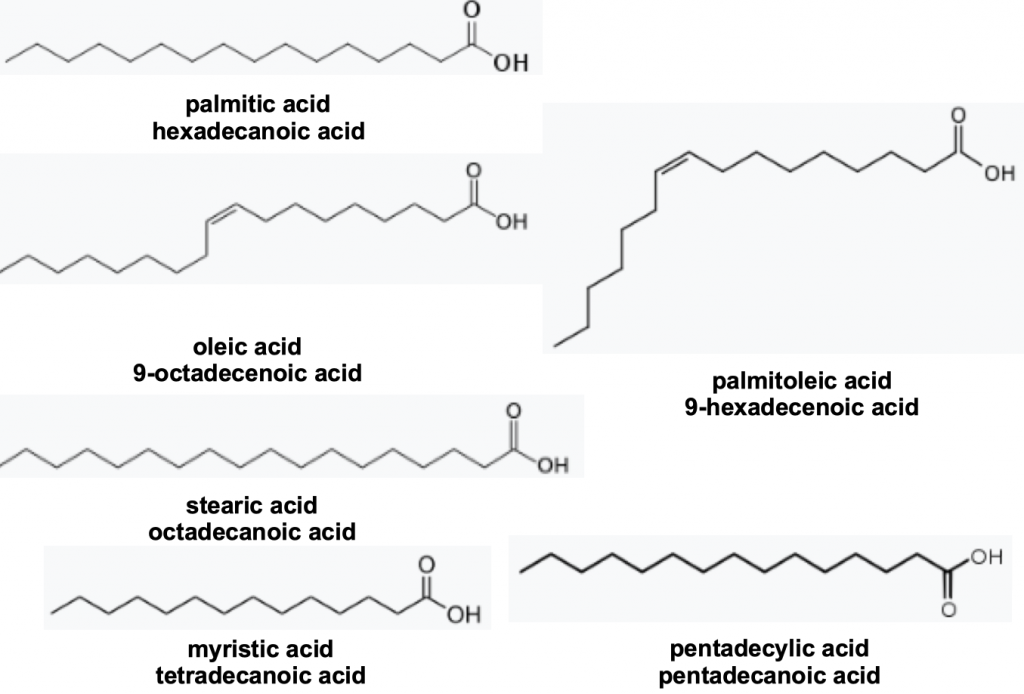
Figure 8 Drone congregation pheromones: palmitic acid 10.5 parts, oleic acid 9.4 parts, stearic acid 3.6 parts, myristic acid 1.7 parts, palmitoleic acid 3 parts, and pentadecylic acid 1 part
Forager pheromone
Ethyl oleate (Figure 9), released by older forager bees, regulates the maturation of of nurse bees to avoid too many house bees becoming foragersxvi.
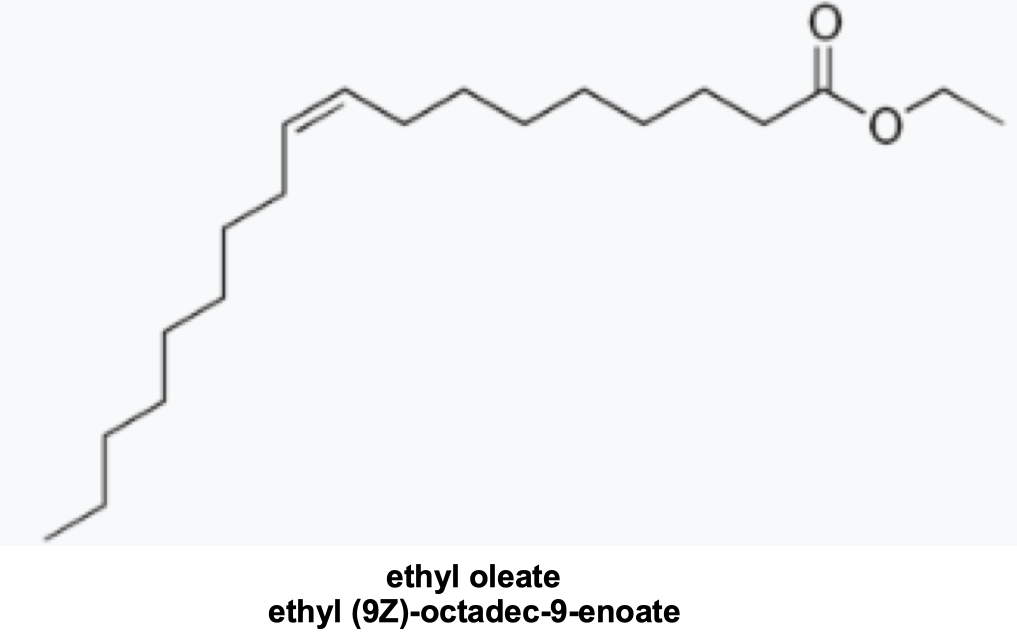
Figure 9 Forager pheromone, a simple ethyl ester of oleic acid
We can expect to learn a whole lot more about chemical communication in bees, not withstanding this limited review. From a practical perspective understanding the role of pheromones and plant volatiles gives us some cues about how we as beekeepers may more sensibly manage bees. Not lunching on bananas, employing smoke to mask chemical signalling amongst bees, recognising that robbing bees are best left alone and experimenting with perfume sprays – as an alternative to employing smoke – on total fire ban days may all be ways to more effective control of bees. And replacing an ailing queen, neither capable of laying well nor communicating well with her offspring, does make a lot of common sense.
Readings
iWikipedia lists twenty nine pricipal components https://en.wikipedia.org/wiki/Lavender_oil:
One group reports some forty individual compounds: Dong, G., Xiaohui Bai, X., Aoken Aimila, A., Aisa, H.A. and Maiwulanjiang, M. (2020). Study on lavender essential oil chemical compositions by GC-MS and improved pGC. Molecules 25(140:3166 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7397202/ doi: 10.3390/molecules25143166
Yet another group reports some seventy eight compunds Smigielski, K., Raj, A.Krosowiak, K. and Gruska, R. (2009). Chemical composition of the essential oil of Lavandula angustifolia cultivated in Poland. Journal of Essential Oil Bearing Plants 12(3):338–347. doi:10.1080/0972060x.2009.10643729
iiNasonov, N.V. (1885). Nasonov pheromone. https://en.wikipedia.org/wiki/Nasonov_pheromone Image at https://commons.wikimedia.org/w/index.php?curid=45861527
iiiPickett, J.A., Williams, I.H., Martin, A.P. and Smith M.C. (1980). The Nasonov pheromone of the honeybee, Apis mellifera L. (Hymenoptera, Apidae), Part I. Chemical characterisation. Journal of Chemical Ecology 6(2):425-434. doi.org/10.1007/BF01402919
Williams, I.H., Pickett, J.A. and Martin, A.P. (198l). Nasonov pheromone of the honeybee, Apis mellifera L. (Hymenoptera, Apidae), Part II. Bioassay of chemical components with foragers. Journal of Chemical Ecology 7(2):225-237. doi:10.1007/BF00995745
Pickett, J.A., Williams, I.H., Smith, M.C. and Martin, A.P. (1981). Nasonov pheromone of the honey bee, Apis mellifera L. (Hymenoptera, Apidae), Part III. Regulation of pheromone composition and production. Journal of Chemical Ecology, 7(3):543–554. doi:10.1007/bf00987702
ivRusty (2017). Honey bee pheromones: common scents. https://www.honeybeesuite.com/honey-bee-pheromones-common-scents/
vKoetz, A. (2013). The Asian honey bee (Apis cerana) and its strains – with special focus on Apis cerana Java genotype, 59pp. Queensland Department of Agriculture, Forestry and Fisheries https://www.planthealthaustralia.com.au/wp-content/uploads/2018/10/Asian-Honey-Bee-Literature-Review.pdf
viNaik, D.G., Gadre, R.V., Kapadi, A.H., Singh, M.K., Suryanarayana, M.C. and Kshirsagar, K.K. (1988). Nasonov gland pheromone of the Indian honeybee, Apis cerana indica. Journal of Apicultural Research 27(4):205-206. doi:10.1080/00218839.1988.1110
viiMatsuyama, S., Suzuki, H. and Sasagawa, H. (2000). Chemical analysis of worker pheromone component of Asian honeybees.v Proceedings of the 4th Asian apiculture association international conference, Kathmandu.
Matsuyama S., Suzuki T. and Sasagawa H. (1997). Semiochemicals in the Japanese honeybee, Apis cerana japonica Rad. Zoological Science 14:49.
viiiButler, C.G., Fletcher, D.J.C. and Watler, D. (1969). Nest-entrance marking with pheromones by the honeybee Apis mellifera L. and by a wasp, Vespula vulgarjs L. Animal Behaviour 17:142–147. doi:10.1016/0003-3472(69)90122-5
ixKastberger, G., Raspotnig, G., Biswas, S. and Winder, O. (2010). Evidence of Nasonov scenting in colony defence of the giant honeybee Apis dorsata. Ethology 104(1):27–37. doi:10.1111/j.1439-0310.1998.tb00027.x
xWinston, M.L. (1987). The biology of the honey bee, pp.131-132. Harvard University Press, 281pp.
xiPickett, J.A., Williams, I.H. and Martin, A.P. (1982). (Z)-11-Eicosen-1-ol, an important new pheromonal component from the sting of the honey bee, Apis mellifera L. (Hymenoptera, Apidae.). Journal of Chemical Ecology 8(1):163–175. doi.org/10.1007/BF00984013
xiiCassier, P., Tel-Zur, D., and Lensky, Y. (1994). The sting sheaths of honey bee workers (Apis mellifera L.). Structure and alarm pheromone secretion. Journal of Insect Physiology 40(1):23–32. doi:10.1016/0022-1910(94)90108-2
xiiiCamargos, A.F., Cossolin, J.F.S., Martínez, L.C., Gonçalves, W.G., dos Santos, M.H., Zanuncio, J.C., and Serrão, J.E. (2020). Morphology and chemical composition of the Koschewnikow gland of the honey bee Apis mellifera (Hymenoptera: Apidae) workers engaged in different tasks. Journal of Apicultural Research 59(5):1037-1048. doi:10.1080/00218839.2020.1736
xivCollison, C. (2022). A closer look: Colony defence — Guarding and stinging. The Australasian Beekeeper 123(8):40-42.
xvVillar, G., Wolfson, M.D., Hefetz, A. and Grozinger, C.M. (2017). Evaluating the role of drone-produced chemical signals in mediating social interactions in honey bees (Apis mellifera). Journal of Chemical Ecology 44(1):1–8. doi:10.1007/s10886-017-0912-2
xviLeoncini, I., Le Conte, Y., Costagliola, G., Plettner, E., Toth, A.L., Wang, M., Huang, Z., Bécard, J.-M., Crauser, D., Slessor, K.N. and Robinson, G.E. (2004). Regulation of behavioral maturation by a primer pheromone produced by adult worker honey bees. Proceedings of the National Academy of Sciences 101(50):17559–17564. doi:10.1073/pnas.0407652101
