
Alan Wade
The Apis dorsata group are the whoppers of the bee kingdom: they have the largest body mass, the longest and largest stings, by far the largest venom glands and, to boot, one extra sting barb. They are also exceptionally aggressive and have distinctive behaviours that ward off the most unwelcome intruder. This accords them the status of being the most savage insect on earth.
It is also instructive to reflect that these giant bees have been around for a long time having a lineage that dates back to the Mid Miocene (Figures 1 and 2). That was some 14-16 million years ago and that was way before primitive forms of Homo sapiens first appeared just 300,000 ybp.
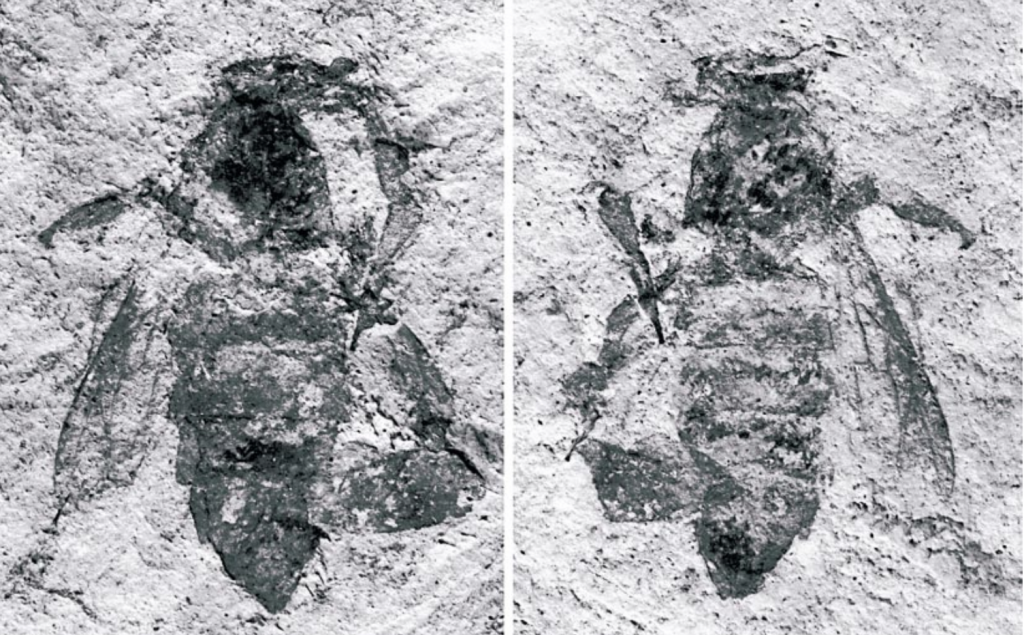
Figure 1 Photomicrographs of Apis lithohermaeai

Figure 2 Evolutionary pathway of the genus Apis (Engle 2006)
Enigmatic giant honey bees
Charles Michener’s Bees of the Worldii lists one hundred and thirty eight scientific names for honey bees. While some are well known to palaeontologists many are extinct. However the majority are simply old names for the ten or maybe twelve different honey bees currently recognised (Table 1).
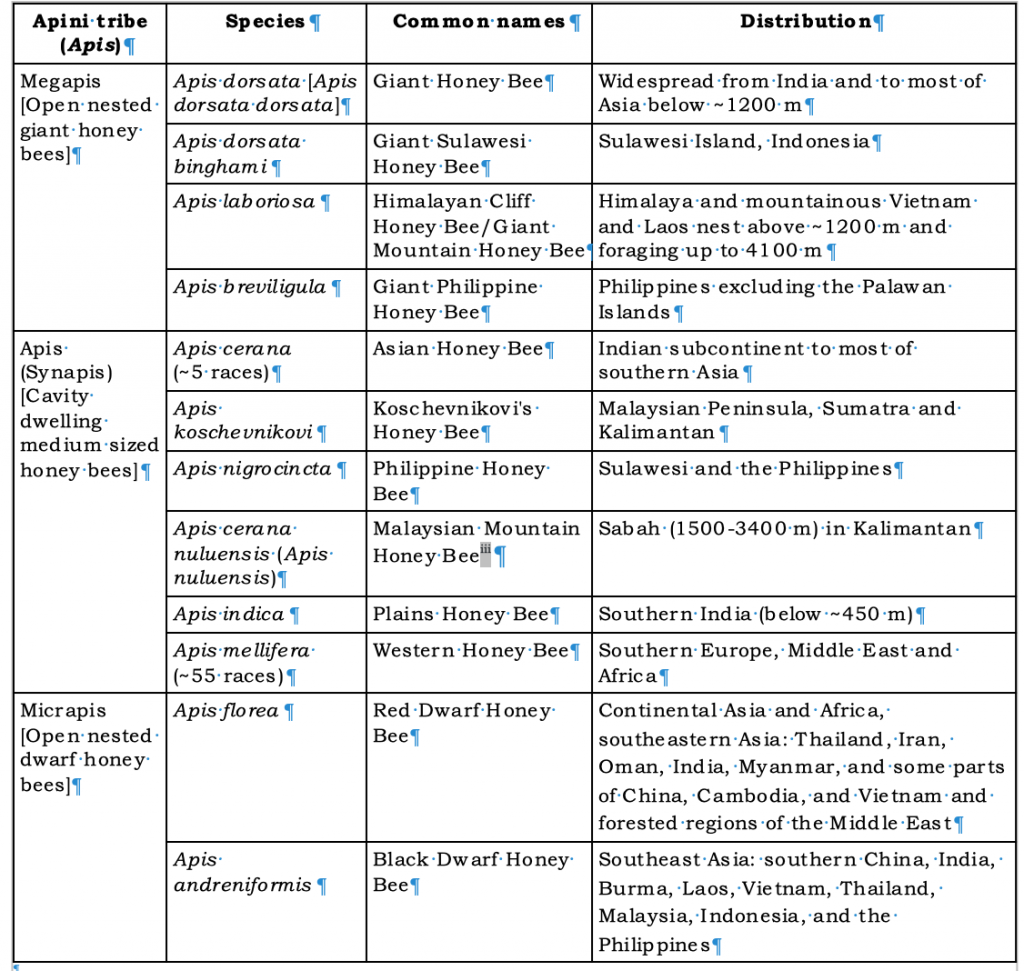
Table 1 Extant (existing) honey bees
*On the basis of detailed morphometric measurements Apis nuluensis, found at high elevation (1500-3400 m) in Sabah, has sometimes been accorded separate species statusiii.
Giant honey bees are widespread in India and Asia but do not extend eastward to Timor, Irian Jaya, Papua New Guinea or to the Pacific (Figure 3).
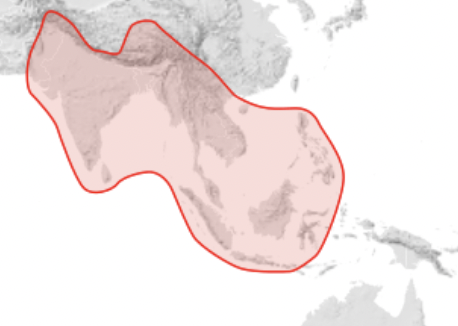
Figure 3 Geographic range of the giant honey bees of the Apis dorsata groupiv
Until relatively recently there was thought to be a single extant giant honey bee, Apis dorsata. It is spread right across the tropical Indian and southern Asian subcontinents. There are now known to be a total of three, perhaps four, different species of giant honey bee. First to be recognised as a new species was the Himalayan Cliff Honey Bee or Giant Mountain Honey Bee, Apis laboriosa. It is well known for its adaptation to seasonal nesting and foraging of summer flora to altitudes up to 4100 m and for its being the largest of all honey bees (Table 2). Then have come more recent genetic studies that has accorded the long isolated Giant Philippine Honey Bee, Apis breviligula, separate species statusv. On the back burner is the also isolated Giant Sulawesi Honey Bee, Apis dorsata binghami. It, like the other two species and Asian mainland Apis dorsata, has different abdomen and stripe patterning but its genetic makeup is not sufficiently clear to determine whether it, too, has been isolated long enough to give it separate species status.
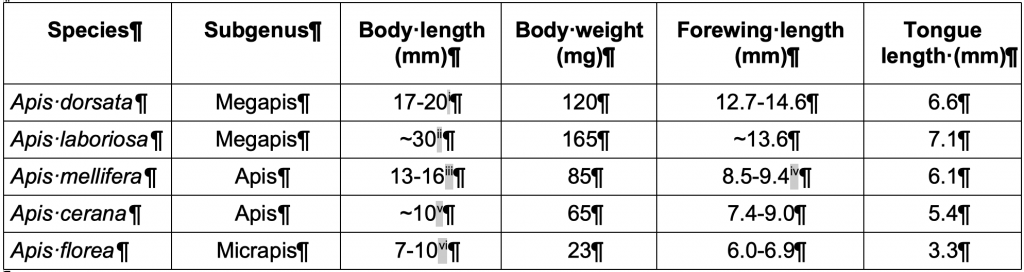
Table 2 Defining morphometric features of representative worker honey bee subgenera (approximate values)xii
Size estimates vary by region: for example Chinese populations of giant honey bee species report smaller length by width forewing measurements for Apis dorsata 12.8 x 4.3 mm and for Apis laboriosa 13.6×4.5 mmxiii.
Other defining features of the giant honey bees are evident at the colony (super organism) level (Table 3) though there are few published studies on the nest and colony characteristics of the giant Sulawesi and Philippine honey bees.
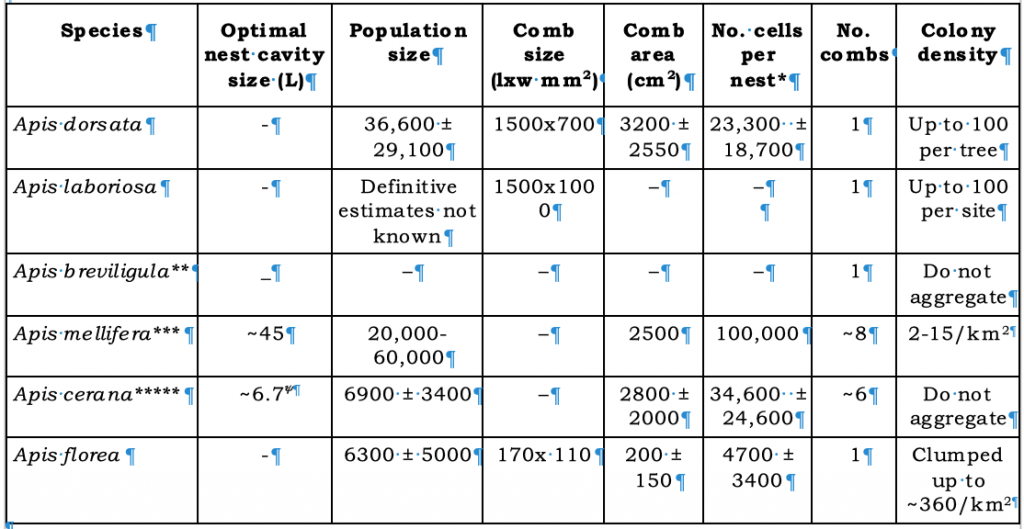
Table 3 Giant honey bee colony population metrics (numbers rounded) compared with other sub generaxiv
*In giant honey bees workers and drones are raised in the same sized cellxv.
**Studies of the biology of the Giant Philippine Honey Bee, Apis breviligula and the Giant honey bee of the Sulawesi, Apis dorsata binghami are few though their biology appears similar to that of widespread Apis dorsata dorsata: Both commonly nest in tall trees, but A. dorsata binghami is less likely to aggregate in large numbers in trees (several to ten for A. dorsatsa binghami and up to around one hundred for Apis dorsata dorsata)xvi
***Apis mellifera colonies are larger and less numerous per square kilometre in Europe than those found in Africa
****Akratanakul,xvii proposes a 20 L super for keeping Apis cerana as opposed to a 40 L box for keeping Apis mellifera bees
Overall the giant honey bees are becoming more widespread in the tropics but are absent from Central America, the Pacific Islands, Australia, New Zealand, Irian Jaya and Papua New Guinea. The giant honey bees are the natural hosts of parasitic mites belonging to the genus Tropilaelapsxviii.
The most defining feature of the different members of the Apis dorsata group is that they are geographically separated where allopatric speciation, genetic drift due to long isolation, has occurred. The overall picture is one of the large geographic range of Apis dorsata (Apis dorsata dorsata) and island occupation by the variants (Figure 4).
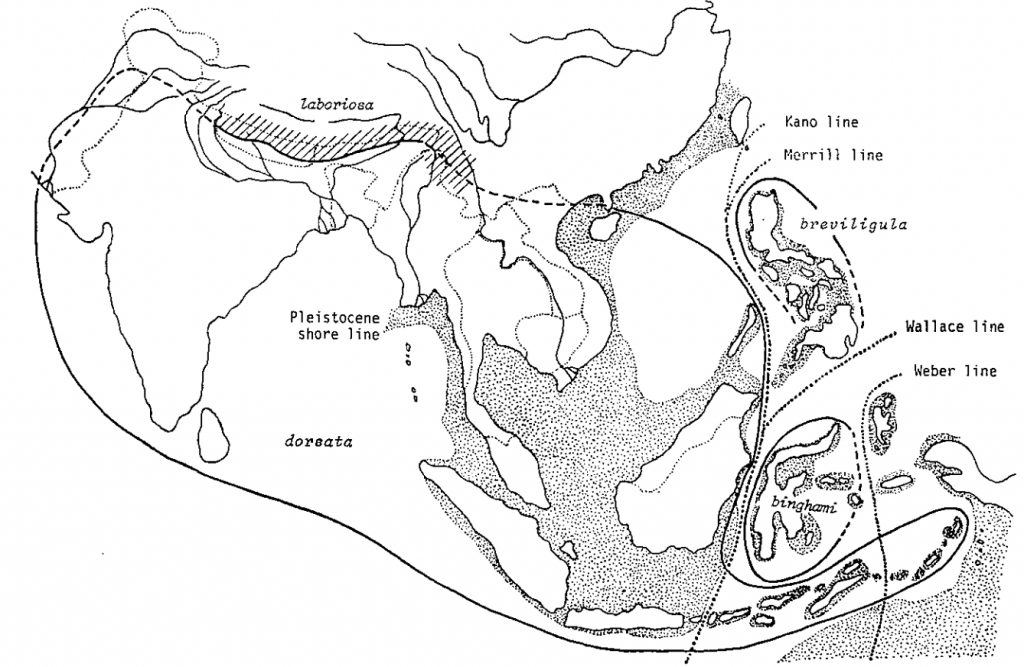
Figure 4 Geographical distribution of four forms of Apis dorsata-group (Megapis)xix
County distribution and association with co-evolved Tropilaelaps parasites is shown below (Table 4).
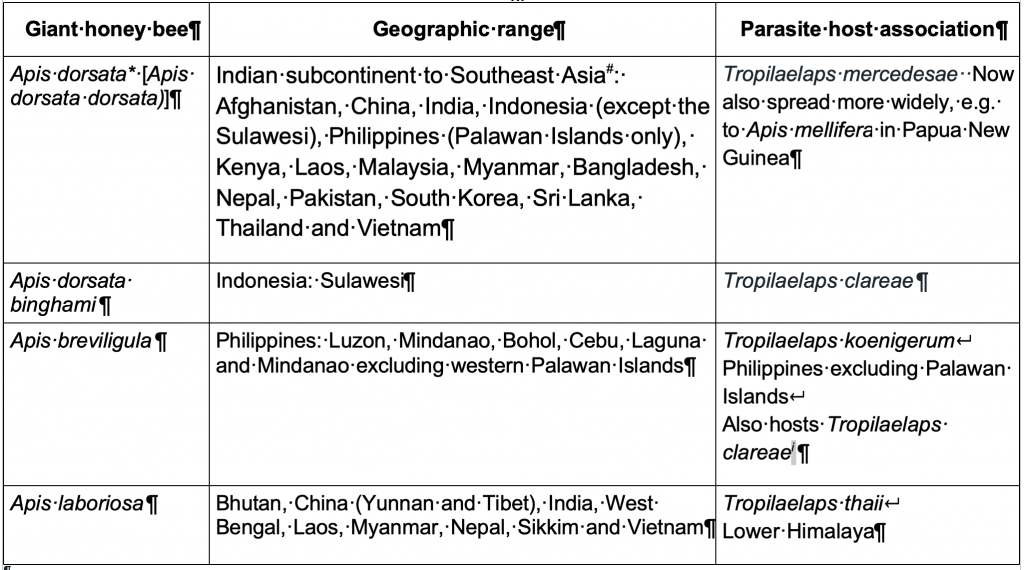
Table 4 Distribution of giant honey bees and associated Tropilaelaps parasites
*Apis dorsata also hosts the external parasites Acarapis externus and Acarapis dorsalis but their origin is uncertain as they also impact on Apis cerana and introduced Apis mellifera. A. externus is found in Australia but is not accorded pest status.
#Apis dorsata has spread east to Kenya and Afghanistan, so may be spreading elsewhere in the Near East.
There is a limited literature on giant honey bee pheromones. We know that Nasonov glands of giant honey bees produce alarm pheromones. In the cavity dwelling Apis sub genus, they function primarily to facilitate bee aggregation, for example forming part of the glue that keeps swarming bees together and that assists bees in orienting themselves at colony entrances.
In a study conducted by Shearer and coworkers, we learn that principle component of queen substance, 9-oxodec-trans-2-enoic acid – we might call it hive glue – is produced in comparable quantities by Apis dorsata, Apis cerana and Apis melliferaxxi queens. This and associated queen mandibular gland pheromones are inimical to the cohesion and functioning of the brood nest and functions in other ways in maintaining colony homeostasis: the roles of queen substance are as disparate as control of drone comb and queen cell construction, control over the production of both supersedure and swarm queens and for queen retinue and swarm cohesion.
The self-assembly processes in honey bees is easily recognised in phenomena such as swarming and when bees cluster together when they are dislodged from frames and capacity to coordinate defence activities in response to hive invasion by predators. Yet the range of behaviours differs distinctly between dwarf, cavity nesting and giant honey bees well illustrated in the disparate strategies for colony self defence adopted by the different honey bee groups.
Colony defence
Apiaries comprising domesticated western and Asian honey bees – European and a few South African races of Apis mellifera and Japanese and Chinese races of Apis cerana – are afforded a large measure of protection by the hive we provide them. All wild colonies of cavity dwelling honey bees gain further protection from more sturdy and better insulated hollows, from their proclivity to employ small defendable colony entrances and from some behavioural traits, notably Asian cavity-dwelling honey bees heat balling insect predators.
To varying degree all honey bees employ stinging to fend off vertebrate predators (bears, mice, people, skunks, gophers…), their venom having much the same potency across all species. However the amount of venom they can deliver and their proclivity to sting varies widely.
The dwarf honey bees employ strategies such as camouflage (hiding nests in leafy undergrowth), applying sticky exudates to branch attachment to repel ants and retreat and readily abscond in response to wholesale predation. Giant honey bee nests, on the other hand, are in the open and quite conspicuous, though typically inaccessible. They build their nests high in large trees (Figure 5) or attach their singe comb to rock overhangs and have a lethal sting apparatus (Table 5).
This all said the giant honey bee defence lies as much in their large sting and venom apparatus as in their aggression behaviour. This makes the giant honey bees such dangerous animals.
Jerzy Woyke and coworkersxxii in assessing the best way to handle giant honey bees noted that:
The extraordinary defense behavior of Apis dorsata is well known. Roepke (1930)xxiii described an A. dorsata attack, during which the worker bees followed him for 500 m. When Lindauer (1956)xxiv was attacked, the bees followed him for 2 km. Near Anuradhapura, Sri Lanka, three water buffalos were killed by A. dorsata. According to Morse and Laigo (1969) [loc. cit.] there is no question that this is the most ferocious stinging insect on earth.
Open nesting bees – both the dwarf and giant bees form a protective barrier at their point of attachment to the substate effectively removing the risk of predation by ants. Other behaviours such as hissing and extending the size of the curtain and bee shimmering, the latter two particularly pronounced in the giant honey bees, are effective in preventing attack by predatory insects such as waspsxxv.

Figure 5 Apis dorsata colony in high canopy of a silk cotton tree at Preah Khan Temple near Siem Reap in Cambodia mid November 2014
Photo: Alan Wade
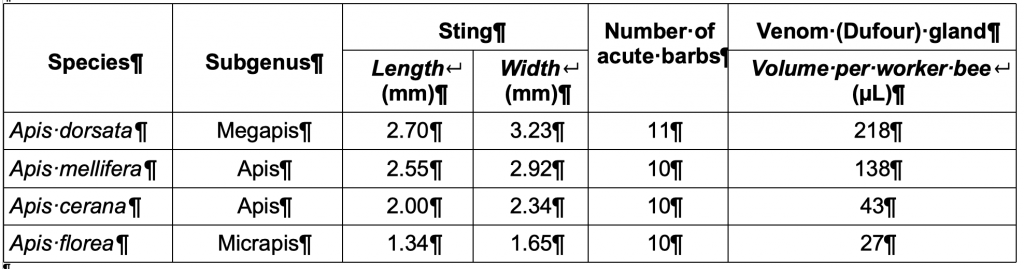
Table 5 Defence characteristics of representative honey bee speciesxxvi
One detail of a behavioural trait in both the Giant Honey Bee and in the Giant Philippine Honey Bee is upward abdomen flipping (the aforementioned shimmering) captured on camera by Kastberger, Weihmann, Zierler and Hötzlxxvii. This behavioural trait helps regulate brood nest temperature of the bee curtain protecting the open nest and would appear effective in warding off wasp predators. Much recommended is viewing of the Kasterberger short video clips demonstrating dorso-ventral abdomen flipping response of Apis dorsata colony to wasps1.
Postscript
To conclude our introduction to these giant bees there is of course a long history of their association with human settlement in southern Asia and the problems they present. A favourite childhood recollection of the strife these creatures inflict is told in the Red Dog yard spun by Rudyard Kipling in his second jungle bookxxviii.
A cautionary tale
When marauding hordes of the Red Hunting Dog or Dholexxix, Cuon alpinus, of the Deccan Plateau descended from the Seeonee Hills they did not reckon on the fearsome giant honey bee (Apis dorsata) festooning the cliffs of the Northern Indian Waingunga River Gorge:
The split and weatherworn rocks of the gorge of the Waingunga had been used since the beginning of the Jungle by the Little People of the Rocks—the busy, furious, black wild bees of India; and, as Mowgli knew well, all trails turned off half a mile before they reached the gorge. For centuries the Little People had hived and swarmed from cleft to cleft, and swarmed again, staining the white marble with stale honey, and made their combs tall and deep in the dark of the inner caves, where neither man nor beast nor fire nor water had ever touched them. The length of the gorge on both sides was hung as it were with black shimmery velvet curtains, and Mowgli sank as he looked, for those were the clotted millions of the sleeping bees…
As he listened he heard more than once the rustle and slide of a honey-loaded comb turning over or falling away somewhere in the dark galleries; then a booming of angry wings, and the sullen drip, drip, drip, of the wasted honey, guttering along till it lipped over some ledge in the open air and sluggishly trickled down on the twigs.
The pursuing Red Dog pack, led by Mowgli, plunged into the gorge:
Overhead they could hear furious short yells that were drowned in a roar like breakers—the roar of the wings of the Little People of the Rocks. Some of the dholes, too, had fallen into the gullies that communicated with the underground caves, and there choked and fought and snapped among the tumbled honeycombs, and at last, borne up, even when they were dead, on the heaving waves of bees beneath them, shot out of some hole in the river-face, to roll over on the black rubbish-heaps.
In the next episode, Part II, we will track the biology of each of the giant-honey bees those thrown up by the complex biogeography of the South China Sea and South Asian mainland. This region is the cauldron of evolution of not only the giant honey bees but also of all honey beesxxx.
1See video clips for Kastberger, Hötzl, Kranner, Weiss, Maurer, and Weihmann (2014) loc. cit. at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4209238/#MOESM2 I suggest you download images from this website to your desktop, then use the open with option in the file menu. I found that the videos would not open in iMovi, the default Apple-Macintosh media player, but was resolved by using the VLC media player.
Readings
iEngle, M.S. (2006). A giant honey bee from the Middle Miocene of Japan (Hymenoptera: Apidae). American Museum Novitates 3504(1):1-12. doi:10.1206/0003-0082(2006)504[0001:AGHBFT]2.0.CO;2
iiMichener, C.D. (2007). The Bees of the World, second edition. The Johns Hopkins University Press, Baltimore, Maryland.
iiiFuchs, S., Koeniger, N. and Tingek, S. (1996). The morphometric position of Apis nuluensis Tingek, Koeniger and Koeniger, 1996 within cavity-nesting honey bees. Apidologie 27(5):397-405 doi:10.1051/apido:19960507 https://www.researchgate.net/publication/248855256_The_morphometric_position_of_Apis_nuluensis_Tingek_Koeniger_and_Koeniger_1996_within_cavity-nesting_honey_bees [accessed May 25 2022
Koeniger N., Koeniger G., Gries M., Tingek S. and Kelitu A. (1996). Reproductive Isolation of A nuluensis, Tingek, Koeniger and Koeniger 1996, by species specific mating time. Apidologie 27(5):353-360. https://hal.archives-ouvertes.fr/hal-00891379/document doi:10.1051/apido:2000125
ivWikimedia Commons Apis dorsata distribution map.svg https://commons.wikimedia.org/w/index.php?curid=13706771
vLo, N., Gloag, R.S., Anderson, D.L. and Oldroyd, B.P. (2010). A molecular phylogeny of the genus Apis suggests that the giant honey bee of the Philippines, A. breviligula Maa, and the Plains Honey Bee of southern India, A. indica Fabricius, are valid species. Systematic Entomology 35(2):226–233. The Royal Entomological Society. doi:10.1111/j.1365-3113.2009.00504.x
viPlant Health Australia (downloaded 15 April 2022). Giant honey bees. https://beeaware.org.au/archive-pest/giant-honey-bees/#ad-image-0
viiGregory, M., and Jack, C. (February 2022). Himalayan giant honey bee, cliff honey bee Apis laboriosa Smith (Insecta: Hymenoptera: Apidae). University of Florida. https://edis.ifas.ufl.edu/publication/IN1348 doi.org/10.32473/edis-IN1348-2022
viiiNew South Wales Department of Primary Industries (downloaded 16 April 2022). European honey bee. https://www.dpi.nsw.gov.au/biosecurity/plant/bees-and-wasps/european-honeybee Note that honey bee sizes vary between season, subspecies type and other factors such as nutrition and that European races of Apis mellifera tend to be larger that those found in Africa.
ixKekeçoğlu, M., Bouga, M., Soysal, M.I. and Harizanis, P. (2007). Morphometrics as a tool for the study of genetic variability of honey bees. Tekirdağ Ziraat Fakültesi Dergisi (Journal of Tekirdag Agricultural Faculty) 4(1):7-15. https://www.researchgate.net/publication/38107507_Morphometrics_as_a_Tool_for_the_Study_of_Genetic_Variability_of_Honey_Bees
Ruttner, F. (1988). Biogeography and taxonomy of honeybees. Springer Verlag, Berlin.
xPlant Health Australia (downloaded 16 April 2022). Asian honey bee. https://beeaware.org.au/archive-pest/asian-honey-bee/#ad-image-0
xiPlant Health Australia (downloaded16 April 2022). Dwarf honey bees. doi:10.1007/978-3-642-72649-1_1
xiiHepburn and Radloff (2011) loc. cit; Cushman, D.A. (19 July 2007). Morphometry http://www.dave- cushman.net/bee/morphometry.html
xiiiCao, L.-F., Zheng, H.-Q., Chen, X., Niu, D.-F., Hu, F.-L. and Hepburn, H.R. (2012). Multivariate morphometric analyses of the giant honey bees, Apis dorsata F. and Apis laboriosa F. in China. Journal of Apicultural Research 51(3):245–251. doi:10.3896/IBRA.1.51.3.05
xivMorse, R.A. and Laigo, F.M. (1969). Apis dorsata in the Philippines (including an annotated bibliography) Laguna, Monograph of Philippine Association of Entomologists 1:1-96. https://www.worldcat.org/title/apis-dorsata-in-the-philippines-including-an-annotated- bibliography/oclc/63374047#similar https://www.amazon.com/Apis-dorsata-Philippines- bibliography-Entomologists/dp/B0007JS7TK
Seeley, T.D., Seeley, R.H. and Akratanakul, P. (1982). Colony defense strategies of the honeybee in Thailand. Ecological Monographs 52(1):43-63. doi.org/10.2307/2937344 https://www.jstor.org/stable/2937344
Seeley, T.D. (2010). Honeybee Democracy, p.57. Princeton University Press.
Moritz, R.F.A., Kraus, F.B., Kryger, P. and Crewe, R.M. (2007). The size of wild honeybee populations (Apis mellifera) and its implications for the conservation of honeybees. Journal of Insect Conservation 11(4):391–397. doi:10.1007/s10841-006-9054-5
Hepburn, R., Duangphakdee, O., Phiancharoen, M. and Radloff, S. (2010). Comb wax salvage by the Red Dwarf Honeybee, Apis florea F. Journal of Insect Behavior 23(2):159–164. doi:10.1007/s10905-010-9205-0
Karlsson, T. (1990). The natural nest of the Asian hive bee (Apis cerana) in Bangladesh: A minor field study. Working paper, International Rural Development Centre, Swedish University of Agricultural Sciences No. 134, 35pp. https://www.cabi.org/ISC/abstract/19910229922
xvChuttong, B., Buawangpong, N. and Burgett, M.D. (2019). Drone production by the giant honey bee Apis dorsata F. (Hymenoptera: Apidae). Sociobiology 66(3):475. doi:10.13102/sociobiology.v66i3.4355 https://www.researchgate.net/publication/337437506_Drone_Production_by_the_Giant_Honey_Bee_Apis_dorsata_F_Hymenoptera_Apidae
xviNagir, M.T., Atmowidi, T. and Kahono, S. (2016). The distribution and nest-site preference of Apis dorsata binghami at Maros Forest, South Sulawesi, Indonesia. Journal of Insect Biodiversity 4(23):1-4. doi:10.12976/jib/2016.4.23 https://www.researchgate.net/publication/311972586_The_distribution_and_nest-site_preference_of_Apis_dorsata_binghami_at_Maros_Forest_South_Sulawesi_Indonesia
xviiAkratanakul, P. (1986) p.43. Beekeeping in Asia, 121pp. Food and Agriculture Services Bulletin 68/4, Rome. https://victoriancollections.net.au/items/5313c4ab2162ef22accd9360
xviiiWade, A. (October 2021). Bee Buzz Box October 2021 – Phoretic honey bee mites. https://actbeekeepers.asn.au/bee-buzz-box-october-2021-phoretic-honey-bee-mites/?
Wade, A. (2022). Phoretic honey bee mites. The Australasian Beekeeper 123(7):14-18.
xixSakagami, S.F., Matsumura, T. and Ito, K. (1980). Apis laboriosa in Himalaya, the little known world largest honeybee (Hymenoptera, Apidae). Insecta Matsumurana 19:47–78. https://eprints.lib.hokudai.ac.jp/dspace/bitstream/2115/9801/1/19_p47-77.pdf
xxAnderson D.L. and Morgan, M.J. (2007). Genetic and morphological variation of bee parasitic Tropilaelaps mites (Acari: Laelapidae): New and re-defined species. Experimental and Applied Acarology 43:1–24. https://link.springer.com/article/10.1007/s10493-007-9103-0 doi:10.1007/s10493-007-9103-0 https://openresearch-repository.anu.edu.au/bitstream/1885/51418/2/01_Anderson_Genetic_and_morphological_2007.pdf
xxiShearer, D.A., Boch, R., Morse, R.A. and Laigo, F.M. (1970). Occurrence of 9-oxodec-trans-2-enoic acid in queens of Apis dorsata, Apis cerana and Apis mellifera. Journal of Insect Physiology 16(7):1437-1441. https://www.sciencedirect.com/science/article/abs/pii/0022191070901423 https://doi.org/10.1016/0022-1910(70)901242-3
xxiiWoyke, J., Wilde, J. and Reddy, C. (February 2004). The gentleness of Apis dorsata verified while investigating brood-cross fostering and hygienic behavior. Proceedings of the seventh Asian Apicultural Association Conference and tenth BEENET Symposium, pp.87-91. https://www.researchgate.net/publication/251570611_2004_The_gentleness_of_Apis_dorsata_verified_while_investigating_brood-cross_fostering_and_hygienic_behavior
xxiiiRoepke, W. (1930). Beobachtungen an Indischen Honigbienen, insbesondere an Apis dorsata F. Meded. LandbHoogesh, 26pp. H. Veenman & Zonen — Wageningen. https://library.wur.nl/WebQuery/wurpubs/fulltext/293809
xxivLindauer, M. (1956) . Über die Verständigung bei indischen Bienen. Zeitschrift für vergleichende Physiologie 8(6):521-557. https://doi.org/10.1007/BF00341108 https://link.springer.com/article/10.1007/BF00341108
xxvFuchs, S. and Tautz, J. (2011). Colony defence and natural enemies, Chapter 17, pp.369-395 in Hepburn, H.R. and Radloff, S.E. (eds) (2011). Honeybees of Asia, 671pp. Springer-Verlag Berlin Heidelberg.
xxviRamya, J. and Rajagopal, D. (2008). Morphology of the sting and its associated glands in four different honey bee species. Journal of Apicultural Research 47(1):46–52. doi:10.1080/00218839.2008.11101422
Hepburn, R. and Radloff, S.E. (Eds) (2011) loc. cit.
xxviiKastberger, G., Weihmann, F., Zierler, M. and Hötzl, T. (2014). Giant honeybees (Apis dorsata) mob wasps away from the nest by directed visual patterns. Naturwissenschaften 101(11):861–873. doi: 10.1007/s00114-014-1220-0
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4209238/#MOESM2 https://link.springer.com/article/10.1007/s00114-014-1220-0
Kastberger, G., Weihmann, F. and, Höetzl, T. (2013). Social waves in giant honeybees (Apis dorsata) elicit nest vibrations. Naturwissenschaften 100(7):595–609. doi: 10.1007/s00114-013-1056-z
Kastberger, G., Hötzl, T., Kranner, I., Weiss, S.E., Maurer, M. and Weihmann, F. (2014). Speeding up social waves: Propagation mechanisms of shimmering in giant honeybees. PLoS One 9(1):e86315. doi: 10.1371/journal.pone.0086315
Seeley, T.D., Seeley, R.H. and Akratanakul, P. (1982) loc. cit.
Oldroyd, B.P. and Wongsiri S. (2006). Asian honey bees, 360pp. Cambridge: Harvard University Press.
Woyke, J., Wilde, J., Wilde, M. and Cervancia, C. (2006). Abdomen flipping of Apis dorsata breviligula worker bees correlated with temperature of nest curtain surface. Apidologie 37(4):501–505. doi: 10.1051/apido:2006032 https://www.apidologie.org/articles/apido/pdf/2006/04/m6040.pdf
xxviiiKipling, R. (29 July 1895). The Second Jungle Book, Red Dog. Macmillan Publishers, London. The 1906 edition, New York, The Century Co. https://ia902704.us.archive.org/35/items/junglebooksecond00kipl/junglebooksecond00kipl.pdf
xxixDhole (downloaded 14 May 2022). https://en.wikipedia.org/wiki/Dhole
xxxWade, A. (2017). A review of the global distribution of honey bees. The Australasian Beekeeper 118(10): 447-452; 118(11):485.
